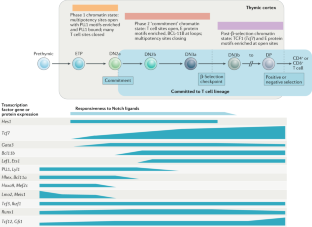
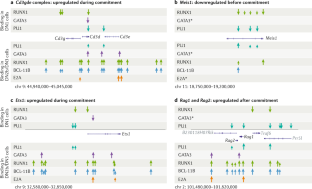
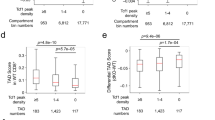
How transcription factors drive choice of the T cell fate
Recent evidence has elucidated how multipotent blood progenitors transform their identities in the thymus and undergo commitment to become T cells. Together with environmental signals, a core group of transcription factors have essential roles in this process by directly activating and repressing specific genes. Many of these transcription factors also function in later T cell development, but control different genes. Here, we review how these transcription factors work to change the activities of specific genomic loci during early intrathymic development to establish T cell lineage identity. We introduce the key regulators and highlight newly emergent insights into the rules that govern their actions. Whole-genome deep sequencing-based analysis has revealed unexpectedly rich relationships between inherited epigenetic states, transcription factor–DNA binding affinity thresholds and influences of given transcription factors on the activities of other factors in the same cells. Together, these mechanisms determine T cell identity and make the lineage choice irreversible.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
206,07 € per year
only 17,17 € per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others

Distinct and temporary-restricted epigenetic mechanisms regulate human αβ and γδ T cell development
Article 27 July 2020

Runx factors launch T cell and innate lymphoid programs via direct and gene network-based mechanisms
Article 10 August 2023
Tcf1 and Lef1 provide constant supervision to mature CD8 + T cell identity and function by organizing genomic architecture
Article Open access 06 October 2021
References
- Hosokawa, H. & Rothenberg, E. V. Cytokines, transcription factors, and the initiation of T-cell development. Cold Spring Harb. Perspect. Biol.10, a028621 (2018). ArticlePubMedPubMed CentralCASGoogle Scholar
- Yui, M. A. & Rothenberg, E. V. Developmental gene networks: a triathlon on the course to T cell identity. Nat. Rev. Immunol.14, 529–545 (2014). ArticleCASPubMedPubMed CentralGoogle Scholar
- Lu, M. et al. The earliest thymic progenitors in adults are restricted to T, NK, and dendritic cell lineage and have a potential to form more diverse TCRβ chains than fetal progenitors. J. Immunol.175, 5848–5856 (2005). ArticleCASPubMedGoogle Scholar
- Singer, A., Adoro, S. & Park, J. H. Lineage fate and intense debate: myths, models and mechanisms of CD4 − versus CD8-lineage choice. Nat. Rev. Immunol.8, 788–801 (2008). ArticleCASPubMedPubMed CentralGoogle Scholar
- Desiderio, S. Temporal and spatial regulatory functions of the V(D)J recombinase. Semin. Immunol.22, 362–369 (2010). ArticleCASPubMedGoogle Scholar
- Rothenberg, E. V., Moore, J. E. & Yui, M. A. Launching the T-cell-lineage developmental programme. Nat. Rev. Immunol.8, 9–21 (2008). ArticleCASPubMedPubMed CentralGoogle Scholar
- Yang, Q., Jeremiah Bell, J. & Bhandoola, A. T-cell lineage determination. Immunol. Rev.238, 12–22 (2010). ArticleCASPubMedPubMed CentralGoogle Scholar
- Romanoski, C. E., Link, V. M., Heinz, S. & Glass, C. K. Exploiting genomics and natural genetic variation to decode macrophage enhancers. Trends Immunol.36, 507–518 (2015). ArticleCASPubMedPubMed CentralGoogle Scholar
- Laiosa, C. V., Stadtfeld, M. & Graf, T. Determinants of lymphoid-myeloid lineage diversification. Annu. Rev. Immunol.24, 705–738 (2006). ArticleCASPubMedGoogle Scholar
- Takahashi, K. & Yamanaka, S. A decade of transcription factor-mediated reprogramming to pluripotency. Nat. Rev. Mol. Cell Biol.17, 183–193 (2016). ArticleCASPubMedGoogle Scholar
- Schebesta, A. et al. Transcription factor Pax5 activates the chromatin of key genes involved in B cell signaling, adhesion, migration, and immune function. Immunity27, 49–63 (2007). ArticleCASPubMedGoogle Scholar
- Lin, Y. C. et al. A global network of transcription factors, involving E2A, EBF1 and Foxo1, that orchestrates B cell fate. Nat. Immunol.11, 635–643 (2010). ArticleCASPubMedPubMed CentralGoogle Scholar
- Seo, W. & Taniuchi, I. Transcriptional regulation of early T-cell development in the thymus. Eur. J. Immunol.46, 531–538 (2016). ArticleCASPubMedGoogle Scholar
- Thompson, P. K. & Zúñiga-Pflücker, J. C. On becoming a T cell, a convergence of factors kick it up a Notch along the way. Semin. Immunol.23, 350–359 (2011). ArticleCASPubMedGoogle Scholar
- Rothenberg, E. V., Ungerback, J. & Champhekar, A. Forging T-lymphocyte identity: intersecting networks of transcriptional control. Adv. Immunol.129, 109–174 (2016). ArticleCASPubMedGoogle Scholar
- Zhou, W. et al. Single-cell analysis reveals regulatory gene expression dynamics leading to lineage commitment in early T cell development. Cell Syst.9, 321–337 e9 (2019). This study focuses on patterns of transcription factor expression in the earliest stages of T cell differentiation and their relationship to developmental potentials at the single-cell level using single-molecule fluorescence in situ hybridization and RNA sequencing for high-sensitivity quantitation of the expression of multiple transcription factor genes in the same single cells. ArticleCASPubMedPubMed CentralGoogle Scholar
- Cleveland, S. M. et al. Lmo2 induces hematopoietic stem cell-like features in T-cell progenitor cells prior to leukemia. Stem Cells31, 882–894 (2013). ArticleCASPubMedPubMed CentralGoogle Scholar
- Hu, G. et al. Transformation of accessible chromatin and 3D nucleome underlies lineage commitment of early T cells. Immunity48, 227–242 e8 (2018). Revealing major changes in chromatin organization during commitment, this study shows that PU.1 motifs predominate in chromatin regions that are open early in pro-T cells. Then PU.1 occupancy gives way to BCL-11B occupancy of open sites across the genome, with evidence that BCL-11B is important as a factor associated with genomic loops in T lineage cells. ArticleCASPubMedPubMed CentralGoogle Scholar
- Ungerbäck, J. et al. Pioneering, chromatin remodeling, and epigenetic constraint in early T-cell gene regulation by SPI1 (PU.1). Genome Res.28, 1508–1519 (2018). This study uses a motif quality metric for a global dissection of how PU.1 functions in open chromatin maintenance, pioneer-like activity and gene regulation in early pro-T cells. It shows that PU.1 respects chromatin constraints but can bind to closed sites if they are of sufficiently high affinity. ArticlePubMedPubMed CentralCASGoogle Scholar
- Johnson, J. L. et al. Lineage-determining transcription factor TCF-1 initiates the epigenetic identity of T cells. Immunity48, 243–257 e10 (2018). This study establishes TCF1 as a controller of open chromatin states in T lineage cells. The authors show enrichment of TCF1 motifs in T cell lineage-associated chromatin sites of two types: one set becoming accessible initially in ETPs, and a larger set becoming accessible around the time of or after commitment. ArticleCASPubMedPubMed CentralGoogle Scholar
- Lavaert, M. et al. Integrated scRNA-Seq identifies human postnatal thymus seeding progenitors and regulatory dynamics of differentiating immature thymocytes. Immunity52, 1088–1104 e6 (2020). ArticleCASPubMedGoogle Scholar
- Le, J. et al. Single-cell RNA-Seq mapping of human thymopoiesis reveals lineage specification trajectories and a commitment spectrum in T cell development. Immunity52, 1105–1118 e9 (2020). Lavaert et al. (2020) and Le et al. provide complementary single-cell transcriptome analyses of pro-T cells in postnatal human thymus. Lavaert et al. focus on potential regulatory network connections, and Le et al. focus on lineage commitment. These two reports elegantly document broad similarities but some differences between early human and mouse stages. ArticleCASPubMedPubMed CentralGoogle Scholar
- Zhang, J. A., Mortazavi, A., Williams, B. A., Wold, B. J. & Rothenberg, E. V. Dynamic transformations of genome-wide epigenetic marking and transcriptional control establish T cell identity. Cell149, 467–482 (2012). ArticleCASPubMedPubMed CentralGoogle Scholar
- Ji, H. et al. Comprehensive methylome map of lineage commitment from haematopoietic progenitors. Nature467, 338–342 (2010). ArticleCASPubMedPubMed CentralGoogle Scholar
- Yoshida, H. et al. The cis-regulatory atlas of the mouse immune system. Cell176, 897–912 e20 (2019). In this comprehensive survey, the authors show that enhancer activity correlates better with gene expression than promoter accessibility and that E protein and TCF family motifs predominate in enhancer sites that become accessible after T cell lineage commitment. A global analysis further relates specific factors to chromatin accessibility changes. ArticleCASPubMedPubMed CentralGoogle Scholar
- Radtke, F. et al. Deficient T cell fate specification in mice with an induced inactivation of Notch1. Immunity10, 547–558 (1999). ArticleCASPubMedGoogle Scholar
- Hozumi, K. et al. Delta-like 4 is indispensable in thymic environment specific for T cell development. J. Exp. Med.205, 2507–2513 (2008). ArticleCASPubMedPubMed CentralGoogle Scholar
- Schmitt, T. M. & Zúñiga-Pflücker, J. C. Induction of T cell development from hematopoietic progenitor cells by Delta-like-1 in vitro. Immunity17, 749–756 (2002). ArticleCASPubMedGoogle Scholar
- Hozumi, K., Abe, N., Chiba, S., Hirai, H. & Habu, S. Active form of Notch members can enforce T lymphopoiesis on lymphoid progenitors in the monolayer culture specific for B cell development. J. Immunol.170, 4973–4979 (2003). ArticleCASPubMedGoogle Scholar
- Bray, S. J. Notch signalling: a simple pathway becomes complex. Nat. Rev. Mol. Cell. Biol.7, 678–689 (2006). ArticleCASPubMedGoogle Scholar
- Yashiro-Ohtani, Y., Ohtani, T. & Pear, W. S. Notch regulation of early thymocyte development. Semin. Immunol.22, 261–269 (2010). ArticleCASPubMedGoogle Scholar
- Ciofani, M. & Zúñiga-Pflücker, J. C. A survival guide to early T cell development. Immunol. Res.34, 117–132 (2006). ArticleCASPubMedGoogle Scholar
- Radtke, F., Macdonald, H. R. & Tacchini-Cottier, F. Regulation of innate and adaptive immunity by Notch. Nat. Rev. Immunol.13, 427–437 (2013). ArticleCASPubMedGoogle Scholar
- Mingueneau, M. et al. The transcriptional landscape of αβ T cell differentiation. Nat. Immunol.14, 619–632 (2013). ArticleCASPubMedPubMed CentralGoogle Scholar
- De Obaldia, M. E. et al. T cell development requires constraint of the myeloid regulator C/EBP-α by the Notch target and transcriptional repressor Hes1. Nat. Immunol.14, 1277–1284 (2013). ArticlePubMedPubMed CentralCASGoogle Scholar
- Wong, G. W., Knowles, G. C., Mak, T. W., Ferrando, A. A. & Zúñiga-Pflücker, J. C. HES1 opposes a PTEN-dependent check on survival, differentiation, and proliferation of TCRβ-selected mouse thymocytes. Blood.120, 1439–1448 (2012). ArticleCASPubMedPubMed CentralGoogle Scholar
- Muñoz-Descalzo, S., de Navascues, J. & Martinez Arias, A. Wnt-Notch signalling: an integrated mechanism regulating transitions between cell states. Bioessays34, 110–118 (2012). ArticlePubMedCASGoogle Scholar
- Chen, E. L. Y., Thompson, P. K. & Zúñiga-Pflücker, J. C. RBPJ-dependent Notch signaling initiates the T cell program in a subset of thymus-seeding progenitors. Nat. Immunol.20, 1456–1468 (2019). The authors develop a mouse strain in which the Notch-interacting transcription factor RBPJ can be deleted and then inducibly restored. The results confirm the need for sustained intrathymic signalling and show, unexpectedly, that a Notch signal via RBPJ needs to be delivered to bone marrow multipotent progenitors even before they reach the thymus. ArticleCASPubMedPubMed CentralGoogle Scholar
- Yu, V. W. et al. Specific bone cells produce DLL4 to generate thymus-seeding progenitors from bone marrow. J. Exp. Med.212, 759–774 (2015). ArticleCASPubMedPubMed CentralGoogle Scholar
- Hirano, K. et al. Delta-like 4-mediated Notch signaling is required for early T-cell development in a three-dimensional thymic structure. Eur. J. Immunol.45, 2252–2262 (2015). ArticleCASPubMedGoogle Scholar
- Wolfer, A., Wilson, A., Nemir, M., MacDonald, H. R. & Radtke, F. Inactivation of Notch1 impairs VDJβ rearrangement and allows pre-TCR-independent survival of early αβ lineage thymocytes. Immunity16, 869–879 (2002). ArticleCASPubMedGoogle Scholar
- Romero-Wolf, M. et al. Notch2 complements Notch1 to mediate inductive signaling that initiates early T cell development. J. Cell Biol.219, e202005093 (2020). ArticleCASPubMedPubMed CentralGoogle Scholar
- Yun, T. J. & Bevan, M. J. Notch-regulated ankyrin-repeat protein inhibits Notch1 signaling: multiple Notch1 signaling pathways involved in T cell development. J. Immunol.170, 5834–5841 (2003). ArticleCASPubMedGoogle Scholar
- Hosoya, T. et al. GATA-3 is required for early T lineage progenitor development. J. Exp. Med.206, 2987–3000 (2009). ArticleCASPubMedPubMed CentralGoogle Scholar
- Germar, K. et al. T-cell factor 1 is a gatekeeper for T-cell specification in response to Notch signaling. Proc. Natl Acad. Sci. USA108, 20060–20065 (2011). ArticleCASPubMedPubMed CentralGoogle Scholar
- Weber, B. N. et al. A critical role for TCF-1 in T-lineage specification and differentiation. Nature476, 63–68 (2011). ArticleCASPubMedPubMed CentralGoogle Scholar
- Del Real, M. M. & Rothenberg, E. V. Architecture of a lymphomyeloid developmental switch controlled by PU.1, Notch and Gata3. Development140, 1207–1219 (2013). ArticlePubMedPubMed CentralCASGoogle Scholar
- Staal, F. J. T. & Sen, J. M. The canonical Wnt signaling pathway plays an important role in lymphopoiesis and hematopoiesis. Eur. J. Immunol.38, 1788–1794 (2008). ArticleCASPubMedPubMed CentralGoogle Scholar
- Xu, Z. et al. Cutting edge: β-catenin-interacting Tcf1 isoforms are essential for thymocyte survival but dispensable for thymic maturation transitions. J. Immunol.198, 3404–3409 (2017). ArticleCASPubMedGoogle Scholar
- Jeannet, G. et al. Long-term, multilineage hematopoiesis occurs in the combined absence of β-catenin and γ-catenin. Blood.111, 142–149 (2008). ArticleCASPubMedGoogle Scholar
- Tiemessen, M. M. et al. The nuclear effector of Wnt-signaling, Tcf1, functions as a T-cell-specific tumor suppressor for development of lymphomas. PLoS Biol.10, e1001430 (2012). ArticleCASPubMedPubMed CentralGoogle Scholar
- Yu, S. et al. The TCF-1 and LEF-1 transcription factors have cooperative and opposing roles in T cell development and malignancy. Immunity.37, 813–826 (2012). ArticleCASPubMedPubMed CentralGoogle Scholar
- Zaret, K. S. & Carroll, J. S. Pioneer transcription factors: establishing competence for gene expression. Genes Dev.25, 2227–2241 (2011). ArticleCASPubMedPubMed CentralGoogle Scholar
- Emmanuel, A. O. et al. TCF-1 and HEB cooperate to establish the epigenetic and transcription profiles of CD4 + CD8 + thymocytes. Nat. Immunol.19, 1366–1378 (2018). The authors demonstrate the close cooperation between TCF1 and the bHLH factor HEB in establishing open chromatin and driving gene expression in DP thymocytes. TCF1 is shown to enhance the binding of HEB to many of its genomic sites and to promote HEB accumulation by blunting Notch signals that otherwise degrade HEB. ArticleCASPubMedPubMed CentralGoogle Scholar
- Frelin, C. et al. GATA-3 regulates the self-renewal of long-term hematopoietic stem cells. Nat. Immunol.14, 1037–1044 (2013). ArticleCASPubMedPubMed CentralGoogle Scholar
- Tindemans, I., Serafini, N., Di Santo, J. P. & Hendriks, R. W. GATA-3 function in innate and adaptive immunity. Immunity.41, 191–206 (2014). ArticleCASPubMedGoogle Scholar
- Hasegawa, S. L. et al. Dosage-dependent rescue of definitive nephrogenesis by a distant Gata3 enhancer. Dev. Biol.301, 568–577 (2007). ArticleCASPubMedGoogle Scholar
- Lim, K. C. et al. Gata3 loss leads to embryonic lethality due to noradrenaline deficiency of the sympathetic nervous system. Nat. Genet.25, 209–212 (2000). ArticleCASPubMedGoogle Scholar
- Kouros-Mehr, H., Slorach, E. M., Sternlicht, M. D. & Werb, Z. GATA-3 maintains the differentiation of the luminal cell fate in the mammary gland. Cell127, 1041–1055 (2006). ArticleCASPubMedPubMed CentralGoogle Scholar
- Van Esch, H. et al. GATA3 haplo-insufficiency causes human HDR syndrome. Nature406, 419–422 (2000). ArticlePubMedCASGoogle Scholar
- Hozumi, K. et al. Notch signaling is necessary for GATA3 function in the initiation of T cell development. Eur. J. Immunol.38, 977–985 (2008). ArticleCASPubMedGoogle Scholar
- Garcia-Ojeda, M. E. et al. GATA-3 promotes T-cell specification by repressing B-cell potential in pro-T cells in mice. Blood121, 1749–1759 (2013). ArticleCASPubMedGoogle Scholar
- Scripture-Adams, D. D. et al. GATA-3 dose-dependent checkpoints in early T cell commitment. J. Immunol.193, 3470–3491 (2014). ArticleCASPubMedGoogle Scholar
- Hosoya, T. et al. Global dynamics of stage-specific transcription factor binding during thymocyte development. Sci. Rep.8, 5605 (2018). ArticlePubMedPubMed CentralCASGoogle Scholar
- Ohmura, S. et al. Lineage-affiliated transcription factors bind the Gata3 Tce1 enhancer to mediate lineage-specific programs. J. Clin. Invest.126, 865–878 (2016). ArticlePubMedPubMed CentralGoogle Scholar
- Xu, W. et al. E2A transcription factors limit expression of Gata3 to facilitate T lymphocyte lineage commitment. Blood121, 1534–1542 (2013). ArticleCASPubMedPubMed CentralGoogle Scholar
- Taghon, T., Yui, M. A. & Rothenberg, E. V. Mast cell lineage diversion of T lineage precursors by the essential T cell transcription factor GATA-3. Nat. Immunol.8, 845–855 (2007). ArticleCASPubMedPubMed CentralGoogle Scholar
- Hosoya, T., Maillard, I. & Engel, J. D. From the cradle to the grave: activities of GATA-3 throughout T-cell development and differentiation. Immunol. Rev.238, 110–125 (2010). ArticleCASPubMedPubMed CentralGoogle Scholar
- Zhu, J. GATA3 regulates the development and functions of innate lymphoid cell subsets at multiple stages. Front. Immunol.8, 1571 (2017). ArticlePubMedPubMed CentralCASGoogle Scholar
- Wei, G. et al. Genome-wide analyses of transcription factor GATA3-mediated gene regulation in distinct T cell types. Immunity35, 299–311 (2011). ArticleCASPubMedPubMed CentralGoogle Scholar
- Nakayama, T. et al. Th2 cells in health and disease. Annu. Rev. Immunol.35, 53–84 (2017). ArticleCASPubMedGoogle Scholar
- Furusawa, J. et al. Critical role of p38 and GATA3 in natural helper cell function. J. Immunol.191, 1818–1826 (2013). ArticleCASPubMedGoogle Scholar
- Hosokawa, H. et al. Methylation of Gata3 protein at Arg-261 regulates transactivation of the Il5 gene in T helper 2 cells. J. Biol. Chem.290, 13095–13103 (2015). ArticleCASPubMedPubMed CentralGoogle Scholar
- Hosokawa, H. et al. Akt1-mediated Gata3 phosphorylation controls the repression of IFNγ in memory-type Th2 cells. Nat. Commun.7, 11289 (2016). ArticleCASPubMedPubMed CentralGoogle Scholar
- Hosokawa, H. et al. Functionally distinct Gata3/Chd4 complexes coordinately establish T helper 2 (Th2) cell identity. Proc. Natl Acad. Sci. USA110, 4691–4696 (2013). ArticleCASPubMedPubMed CentralGoogle Scholar
- Yamagata, T. et al. Acetylation of GATA-3 affects T-cell survival and homing to secondary lymphoid organs. EMBO J.19, 4676–4687 (2000). ArticleCASPubMedPubMed CentralGoogle Scholar
- Buono, M. et al. A dynamic niche provides Kit ligand in a stage-specific manner to the earliest thymocyte progenitors. Nat. Cell Biol.18, 157–167 (2016). ArticleCASPubMedPubMed CentralGoogle Scholar
- Semerad, C. L., Mercer, E. M., Inlay, M. A., Weissman, I. L. & Murre, C. E2A proteins maintain the hematopoietic stem cell pool and promote the maturation of myelolymphoid and myeloerythroid progenitors. Proc. Natl Acad. Sci. USA106, 1930–1935 (2009). ArticleCASPubMedPubMed CentralGoogle Scholar
- Yang, Q. et al. E47 controls the developmental integrity and cell cycle quiescence of multipotential hematopoietic progenitors. J. Immunol.181, 5885–5894 (2008). ArticleCASPubMedGoogle Scholar
- Dias, S., Mansson, R., Gurbuxani, S., Sigvardsson, M. & Kee, B. L. E2A proteins promote development of lymphoid-primed multipotent progenitors. Immunity29, 217–227 (2008). ArticleCASPubMedPubMed CentralGoogle Scholar
- Miyazaki, M. et al. The E-Id protein axis specifies adaptive lymphoid cell identity and suppresses thymic innate lymphoid cell development. Immunity46, 818–834 e4 (2017). E protein activity in ILCs is tightly inhibited by ID2. This study shows that inappropriate expression ofId2or deletion of bothTcf3andTcf12induces abnormal development of ILCs in the thymus. Thus, ID2 acts primarily through E protein neutralization to segregate T cells and ILCs. ArticleCASPubMedPubMed CentralGoogle Scholar
- Murre, C. Helix-loop-helix proteins and the advent of cellular diversity: 30 years of discovery. Genes Dev.33, 6–25 (2019). ArticleCASPubMedPubMed CentralGoogle Scholar
- Braunstein, M. & Anderson, M. K. HEB in the spotlight: transcriptional regulation of T-cell specification, commitment, and developmental plasticity. Clin. Dev. Immunol.2012, 678705 (2012). ArticlePubMedPubMed CentralCASGoogle Scholar
- Bain, G. et al. E2A deficiency leads to abnormalities in αβ T-cell development and to rapid development of T-cell lymphomas. Mol. Cell Biol.17, 4782–4791 (1997). ArticleCASPubMedPubMed CentralGoogle Scholar
- Wojciechowski, J., Lai, A., Kondo, M. & Zhuang, Y. E2A and HEB are required to block thymocyte proliferation prior to pre-TCR expression. J. Immunol.178, 5717–5726 (2007). ArticleCASPubMedGoogle Scholar
- Ikawa, T., Kawamoto, H., Goldrath, A. W. & Murre, C. E proteins and notch signaling cooperate to promote T cell lineage specification and commitment. J. Exp. Med.203, 1329–1342 (2006). ArticleCASPubMedPubMed CentralGoogle Scholar
- Pereira de Sousa, A. et al. Inhibitors of DNA binding proteins restrict T cell potential by repressing Notch1 expression in Flt3-negative common lymphoid progenitors. J. Immunol.189, 3822–3830 (2012). ArticleCASPubMedGoogle Scholar
- Yashiro-Ohtani, Y. et al. Pre-TCR signaling inactivates Notch1 transcription by antagonizing E2A. Genes Dev.23, 1665–1676 (2009). ArticleCASPubMedPubMed CentralGoogle Scholar
- Zook, E. C. & Kee, B. L. Development of innate lymphoid cells. Nat. Immunol.17, 775–782 (2016). ArticleCASPubMedGoogle Scholar
- Moro, K. et al. Innate production of TH2 cytokines by adipose tissue-associated c-Kit + Sca-1 + lymphoid cells. Nature463, 540–544 (2010). ArticleCASPubMedGoogle Scholar
- Koues, O. I. et al. Distinct gene regulatory pathways for human innate versus adaptive lymphoid cells. Cell165, 1134–1146 (2016). ArticleCASPubMedPubMed CentralGoogle Scholar
- Boos, M. D., Yokota, Y., Eberl, G. & Kee, B. L. Mature natural killer cell and lymphoid tissue-inducing cell development requires Id2-mediated suppression of E protein activity. J. Exp. Med.204, 1119–1130 (2007). ArticleCASPubMedPubMed CentralGoogle Scholar
- Hosokawa, H. et al. Bcl11b sets pro-T cell fate by site-specific cofactor recruitment and by repressing Id2 and Zbtb16. Nat. Immunol.19, 1427–1440 (2018). This study reveals the molecular basis of both direct and indirect BCL-11B actions to promote T cell identity and to avoid alternative lineage phenotypes in the thymus. Among the BCL-11B-repressed targets, Id2andZbtb16are shown to be functionally important to control distinct alternative programmes. ArticleCASPubMedPubMed CentralGoogle Scholar
- Ikawa, T., Fujimoto, S., Kawamoto, H., Katsura, Y. & Yokota, Y. Commitment to natural killer cells requires the helix-loop-helix inhibitor Id2. Proc. Natl Acad. Sci. USA98, 5164–5169 (2001). ArticleCASPubMedPubMed CentralGoogle Scholar
- Zook, E. C. et al. Transcription factor ID2 prevents E proteins from enforcing a naive T lymphocyte gene program during NK cell development. Sci. Immunol.3, eaao2139 (2018). ArticlePubMedPubMed CentralGoogle Scholar
- Miyazaki, M. et al. The opposing roles of the transcription factor E2A and its antagonist Id3 that orchestrate and enforce the naive fate of T cells. Nat. Immunol.12, 992–1001 (2011). ArticleCASPubMedPubMed CentralGoogle Scholar
- Jones, M. E. & Zhuang, Y. Stage-specific functions of E-proteins at the β-selection and T-cell receptor checkpoints during thymocyte development. Immunol. Res.49, 202–215 (2011). ArticleCASPubMedGoogle Scholar
- Jones, M. E. & Zhuang, Y. Regulation of V(D)J recombination by E-protein transcription factors. Adv. Exp. Med. Biol.650, 148–156 (2009). ArticleCASPubMedGoogle Scholar
- Jones, M. E. & Zhuang, Y. Acquisition of a functional T cell receptor during T lymphocyte development is enforced by HEB and E2A transcription factors. Immunity27, 860–870 (2007). ArticleCASPubMedPubMed CentralGoogle Scholar
- Bain, G. et al. Regulation of the helix-loop-helix proteins, E2A and Id3, by the Ras-ERK MAPK cascade. Nat. Immunol.2, 165–171 (2001). ArticleCASPubMedGoogle Scholar
- Jones-Mason, M. E. et al. E protein transcription factors are required for the development of CD4 + lineage T cells. Immunity36, 348–361 (2012). ArticleCASPubMedPubMed CentralGoogle Scholar
- Phelan, J. D. et al. Growth factor independent-1 maintains Notch1-dependent transcriptional programming of lymphoid precursors. PLoS Genet.9, e1003713 (2013). ArticleCASPubMedPubMed CentralGoogle Scholar
- Emambokus, N. et al. Progression through key stages of haemopoiesis is dependent on distinct threshold levels of c-Myb. EMBO J.22, 4478–4488 (2003). ArticleCASPubMedPubMed CentralGoogle Scholar
- Allen, R. D. III, Bender, T. P. & Siu, G. c-Myb is essential for early T cell development. Genes Dev.13, 1073–1078 (1999). ArticleCASPubMedPubMed CentralGoogle Scholar
- Georgopoulos, K. The making of a lymphocyte: the choice among disparate cell fates and the IKAROS enigma. Genes Dev.31, 439–450 (2017). ArticleCASPubMedPubMed CentralGoogle Scholar
- Ebihara, T., Seo, W. & Taniuchi, I. Roles of RUNX complexes in immune cell development. Adv. Exp. Med. Biol.962, 395–413 (2017). ArticleCASPubMedGoogle Scholar
- Guo, Y., Maillard, I., Chakraborti, S., Rothenberg, E. V. & Speck, N. A. Core binding factors are necessary for natural killer cell development, and cooperate with Notch signaling during T cell specification. Blood112, 480–492 (2008). ArticleCASPubMedPubMed CentralGoogle Scholar
- Geimer Le Lay, A. S. et al. The tumor suppressor Ikaros shapes the repertoire of Notch target genes in T cells. Sci. Signal.7, ra28 (2014). ArticlePubMedCASGoogle Scholar
- Winandy, S., Wu, L., Wang, J. H. & Georgopoulos, K. Pre-T cell receptor (TCR) and TCR-controlled checkpoints in T cell differentiation are set by Ikaros. J. Exp. Med.190, 1039–1048 (1999). ArticleCASPubMedPubMed CentralGoogle Scholar
- Arenzana, T. L., Schjerven, H. & Smale, S. T. Regulation of gene expression dynamics during developmental transitions by the Ikaros transcription factor. Genes Dev.29, 1801–1816 (2015). ArticleCASPubMedPubMed CentralGoogle Scholar
- Hosokawa, H. et al. Transcription factor PU.1 represses and activates gene expression in early T cells by redirecting partner transcription factor binding. Immunity48, 1119–1134 e7 (2018). PU.1 directly activates its target genes in part by recruiting RUNX1 and SATB1. The result is to ‘steal’ RUNX1 and SATB1 from sites associated with T cell lineage-related genes, thus repressing some of these genes, indirectly, without direct DNA binding. ArticleCASPubMedPubMed CentralGoogle Scholar
- David-Fung, E. S. et al. Transcription factor expression dynamics of early T-lymphocyte specification and commitment. Dev. Biol.325, 444–467 (2009). ArticleCASPubMedGoogle Scholar
- Talebian, L. et al. T-lymphoid, megakaryocyte, and granulocyte development are sensitive to decreases in CBFβ dosage. Blood109, 11–21 (2007). ArticleCASPubMedPubMed CentralGoogle Scholar
- Cortes, M., Wong, E., Koipally, J. & Georgopoulos, K. Control of lymphocyte development by the Ikaros gene family. Curr. Opin. Biol.11, 167–171 (1999). CASGoogle Scholar
- Liu, P., Li, P. & Burke, S. Critical roles of Bcl11b in T-cell development and maintenance of T-cell identity. Immunol. Rev.238, 138–149 (2010). ArticleCASPubMedGoogle Scholar
- Kominami, R. Role of the transcription factor Bcl11b in development and lymphomagenesis. Proc. Jpn. Acad. Ser. B Phys. Biol. Sci.88, 72–87 (2012). ArticleCASPubMedPubMed CentralGoogle Scholar
- Kastner, P. et al. Bcl11b represses a mature T-cell gene expression program in immature CD4 + CD8 + thymocytes. Eur. J. Immunol.40, 2143–2154 (2010). ArticleCASPubMedPubMed CentralGoogle Scholar
- Li, L., Leid, M. & Rothenberg, E. V. An early T cell lineage commitment checkpoint dependent on the transcription factor Bcl11b. Science329, 89–93 (2010). ArticleCASPubMedPubMed CentralGoogle Scholar
- Shibata, K. et al. IFN-γ-producing and IL-17-producing γδ T cells differentiate at distinct developmental stages in murine fetal thymus. J. Immunol.192, 2210–2218 (2014). ArticleCASPubMedGoogle Scholar
- De Obaldia, M. E. & Bhandoola, A. Transcriptional regulation of innate and adaptive lymphocyte lineages. Annu. Rev. Immunol.33, 607–642 (2015). ArticlePubMedCASGoogle Scholar
- Avram, D. & Califano, D. The multifaceted roles of Bcl11b in thymic and peripheral T cells: impact on immune diseases. J. Immunol.193, 2059–2065 (2014). ArticleCASPubMedGoogle Scholar
- Fang, D. et al. Bcl11b, a novel GATA3-interacting protein, suppresses Th1 while limiting Th2 cell differentiation. J. Exp. Med.215, 1449–1462 (2018). ArticleCASPubMedPubMed CentralGoogle Scholar
- Punwani, D. et al. Multisystem anomalies in severe combined immunodeficiency with mutant BCL11B. N. Engl. J. Med.375, 2165–2176 (2016). ArticleCASPubMedPubMed CentralGoogle Scholar
- Hirose, S. et al. Bcl11b prevents the intrathymic development of innate CD8 T cells in a cell intrinsic manner. Int. Immunol.27, 205–215 (2015). ArticleCASPubMedGoogle Scholar
- Kojo, S. et al. Priming of lineage-specifying genes by Bcl11b is required for lineage choice in post-selection thymocytes. Nat. Commun.8, 702 (2017). ArticlePubMedPubMed CentralCASGoogle Scholar
- Longabaugh, W. J. R. et al. Bcl11b and combinatorial resolution of cell fate in the T-cell gene regulatory network. Proc. Natl Acad. Sci. USA114, 5800–5807 (2017). ArticleCASPubMedPubMed CentralGoogle Scholar
- Ikawa, T. et al. An essential developmental checkpoint for production of the T cell lineage. Science329, 93–96 (2010). ArticleCASPubMedGoogle Scholar
- Li, P. et al. Reprogramming of T cells to natural killer-like cells upon Bcl11b deletion. Science329, 85–89 (2010). ArticleCASPubMedPubMed CentralGoogle Scholar
- Kadoch, C. & Crabtree, G. R. Mammalian SWI/SNF chromatin remodeling complexes and cancer: Mechanistic insights gained from human genomics. Sci. Adv.1, e1500447 (2015). ArticlePubMedPubMed CentralCASGoogle Scholar
- Cismasiu, V. B. et al. BCL11B functionally associates with the NuRD complex in T lymphocytes to repress targeted promoter. Oncogene24, 6753–6764 (2005). ArticleCASPubMedGoogle Scholar
- Hosokawa, H. et al. Cell type-specific actions of Bcl11b in early T-lineage and group 2 innate lymphoid cells. J. Exp. Med.217, e20190972 (2020). Bcl11bis expressed in pro-T cells and ILC2s. Id2is an important repressed target gene of BCL-11B in pro-T cells, whereasId2must be co-expressed withBcl11bin ILC2s. This study show that BCL-11B regulates distinct sets of genes in pro-T cells and ILC2s by organizing distinct protein complexes and binding to different genomic regions. ArticlePubMedCASGoogle Scholar
- Ng, K. K. et al. A stochastic epigenetic switch controls the dynamics of T-cell lineage commitment. eLife7, e37851 (2018). This article shows thatcis-acting epigenetic constraints cause delays of the order of days in the activation ofBcl11bfor T cell lineage commitment. The authors establish two-colourBcl11breporter mice to monitor the activation status and enhancer dependence ofBcl11balleles separately. ArticlePubMedPubMed CentralGoogle Scholar
- Li, L. et al. A far downstream enhancer for murine Bcl11b controls its T-cell specific expression. Blood122, 902–911 (2013). ArticleCASPubMedPubMed CentralGoogle Scholar
- Rothenberg, E. V. Dynamic control of the T-cell specification gene regulatory network. Curr. Opin. Syst. Biol.18, 62–76 (2019). ArticlePubMedPubMed CentralGoogle Scholar
- Champhekar, A. et al. Regulation of early T-lineage gene expression and developmental progression by the progenitor cell transcription factor PU.1. Genes Dev.29, 832–848 (2015). ArticleCASPubMedPubMed CentralGoogle Scholar
- Rothenberg, E. V., Hosokawa, H. & Ungerback, J. Mechanisms of action of hematopoietic transcription factor PU.1 in initiation of T-cell development. Front. Immunol.10, 228 (2019). ArticleCASPubMedPubMed CentralGoogle Scholar
- Kawamoto, H. et al. Extensive proliferation of T cell lineage-restricted progenitors in the thymus: an essential process for clonal expression of diverse T cell receptor β chains. Eur. J. Immunol.33, 606–615 (2003). ArticleCASPubMedGoogle Scholar
- Scott, E. W., Simon, M. C., Anastasi, J. & Singh, H. Requirement of transcription factor PU.1 in the development of multiple hematopoietic lineages. Science265, 1573–1577 (1994). ArticleCASPubMedGoogle Scholar
- Kueh, H. Y., Champhekar, A., Nutt, S. L., Elowitz, M. B. & Rothenberg, E. V. Positive feedback between PU.1 and the cell cycle controls myeloid differentiation. Science341, 670–673 (2013). ArticleCASPubMedPubMed CentralGoogle Scholar
- Heinz, S. et al. Effect of natural genetic variation on enhancer selection and function. Nature503, 487–492 (2013). ArticleCASPubMedPubMed CentralGoogle Scholar
- Heinz, S. et al. Simple combinations of lineage-determining transcription factors prime cis-regulatory elements required for macrophage and B cell identities. Mol. Cell38, 576–589 (2010). ArticleCASPubMedPubMed CentralGoogle Scholar
- Ostuni, R. et al. Latent enhancers activated by stimulation in differentiated cells. Cell152, 157–171 (2013). ArticleCASPubMedGoogle Scholar
- Escalante, C. R. et al. Crystal structure of PU.1/IRF-4/DNA ternary complex. Mol. Cell10, 1097–1105 (2002). ArticleCASPubMedGoogle Scholar
- Minderjahn, J. et al. Mechanisms governing the pioneering and redistribution capabilities of the non-classical pioneer PU.1. Nat. Commun.11, 402 (2020). ArticleCASPubMedPubMed CentralGoogle Scholar
- Carotta, S. et al. The transcription factor PU.1 controls dendritic cell development and Flt3 cytokine receptor expression in a dose-dependent manner. Immunity32, 628–641 (2010). ArticleCASPubMedGoogle Scholar
- Seki, M. et al. Recurrent SPI1 (PU.1) fusions in high-risk pediatric T cell acute lymphoblastic leukemia. Nat. Genet.49, 1274–1281 (2017). ArticleCASPubMedGoogle Scholar
- Rosenbauer, F. et al. Lymphoid cell growth and transformation are suppressed by a key regulatory element of the gene encoding PU.1. Nat. Genet.38, 27–37 (2006). ArticleCASPubMedGoogle Scholar
- Franco, C. B. et al. Notch/Delta signaling constrains reengineering of pro-T cells by PU.1. Proc. Natl Acad. Sci. USA103, 11993–11998 (2006). ArticleCASPubMedPubMed CentralGoogle Scholar
- Laiosa, C. V., Stadtfeld, M., Xie, H., de Andres-Aguayo, L. & Graf, T. Reprogramming of committed T cell progenitors to macrophages and dendritic cells by C/EBPα and PU.1 transcription factors. Immunity25, 731–744 (2006). ArticleCASPubMedGoogle Scholar
- Yu, Y. et al. Bcl11a is essential for lymphoid development and negatively regulates p53. J. Exp. Med.209, 2467–2483 (2012). ArticleCASPubMedPubMed CentralGoogle Scholar
- Zohren, F. et al. The transcription factor Lyl-1 regulates lymphoid specification and the maintenance of early T lineage progenitors. Nat. Immunol.13, 761–769 (2012). ArticleCASPubMedPubMed CentralGoogle Scholar
- Yui, M. A., Feng, N. & Rothenberg, E. V. Fine-scale staging of T cell lineage commitment in adult mouse thymus. J. Immunol.185, 284–293 (2010). ArticleCASPubMedGoogle Scholar
- Masuda, K. et al. T cell lineage determination precedes the initiation of TCR β gene rearrangement. J. Immunol.179, 3699–3706 (2007). ArticleCASPubMedGoogle Scholar
- Bell, J. J. & Bhandoola, A. The earliest thymic progenitors for T cells possess myeloid lineage potential. Nature452, 764–767 (2008). ArticleCASPubMedGoogle Scholar
- Wada, H. et al. Adult T-cell progenitors retain myeloid potential. Nature452, 768–772 (2008). ArticleCASPubMedGoogle Scholar
- De Obaldia, M. E., Bell, J. J. & Bhandoola, A. Early T-cell progenitors are the major granulocyte precursors in the adult mouse thymus. Blood121, 64–71 (2013). ArticlePubMedPubMed CentralCASGoogle Scholar
- Zarnegar, M. A., Chen, J. & Rothenberg, E. V. Cell-type-specific activation and repression of PU.1 by a complex of discrete, functionally specialized cis-regulatory elements. Mol. Cell. Biol.30, 4922–4939 (2010). ArticleCASPubMedPubMed CentralGoogle Scholar
- Huang, G. et al. PU.1 is a major downstream target of AML1 (RUNX1) in adult mouse hematopoiesis. Nat. Genet.40, 51–60 (2008). ArticleCASPubMedGoogle Scholar
- Kueh, H. Y. et al. Asynchronous combinatorial action of four regulatory factors activates Bcl11b for T cell commitment. Nat. Immunol.17, 956–965 (2016). ArticleCASPubMedPubMed CentralGoogle Scholar
- Rothenberg, E. V. Programming for T-lymphocyte fates: modularity and mechanisms. Genes Dev.33, 1117–1135 (2019). ArticleCASPubMedPubMed CentralGoogle Scholar
- Isoda, T. et al. Non-coding transcription instructs chromatin folding and compartmentalization to dictate enhancer-promoter communication and T cell fate. Cell171, 103–119 e18 (2017). The authors focus on the distal superenhancer region ofBcl11band show that it harbours a functionally important long non-coding RNA that acts positively on gene regulation. The authors find a severe, allele-specific loss ofBcl11bexpression when long non-coding RNA transcription is truncated. ArticleCASPubMedPubMed CentralGoogle Scholar
- Manesso, E., Chickarmane, V., Kueh, H. Y., Rothenberg, E. V. & Peterson, C. Computational modelling of T-cell formation kinetics: output regulated by initial proliferation-linked deferral of developmental competence. J. R. Soc. Interface10, 20120774 (2013). ArticlePubMedPubMed CentralGoogle Scholar
- Krueger, A., Ziętara, N. & Łyszkiewicz, M. T cell development by the numbers. Trends Immunol.38, 128–139 (2017). ArticleCASPubMedGoogle Scholar
- Cumano, A. et al. New molecular insights into immune cell development. Annu. Rev. Immunol.37, 497–519 (2019). ArticleCASPubMedGoogle Scholar
- Casero, D. et al. Long non-coding RNA profiling of human lymphoid progenitor cells reveals transcriptional divergence of B cell and T cell lineages. Nat. Immunol.16, 1282–1291 (2015). ArticleCASPubMedPubMed CentralGoogle Scholar
- Ha, V. L. et al. The T-ALL related gene BCL11B regulates the initial stages of human T-cell differentiation. Leukemia31, 2503–2514 (2017). ArticleCASPubMedPubMed CentralGoogle Scholar
- Canté-Barrett, K. et al. Loss of CD44 dim expression from early progenitor cells marks T-cell lineage commitment in the human thymus. Front. Immunol.8, 32 (2017). ArticlePubMedPubMed CentralCASGoogle Scholar
- Garvie, C. W., Pufall, M. A., Graves, B. J. & Wolberger, C. Structural analysis of the autoinhibition of Ets-1 and its role in protein partnerships. J. Biol. Chem.277, 45529–45536 (2002). ArticleCASPubMedGoogle Scholar
- Pufall, M. A. & Graves, B. J. Ets-1 flips for new partner Pax-5. Structure10, 11–14 (2002). ArticleCASPubMedGoogle Scholar
- Soufi, A., Donahue, G. & Zaret, K. S. Facilitators and impediments of the pluripotency reprogramming factors’ initial engagement with the genome. Cell151, 994–1004 (2012). ArticleCASPubMedPubMed CentralGoogle Scholar
- Iwafuchi-Doi, M. & Zaret, K. S. Cell fate control by pioneer transcription factors. Development143, 1833–1837 (2016). ArticleCASPubMedPubMed CentralGoogle Scholar
- Chronis, C. et al. Cooperative binding of transcription factors orchestrates reprogramming. Cell168, 442–459 (2017). ArticleCASPubMedPubMed CentralGoogle Scholar
- Weikum, E. R., Knuesel, M. T., Ortlund, E. A. & Yamamoto, K. R. Glucocorticoid receptor control of transcription: precision and plasticity via allostery. Nat. Rev. Mol. Cell. Biol.18, 159–174 (2017). ArticleCASPubMedPubMed CentralGoogle Scholar
- Luna-Zurita, L. et al. Complex interdependence regulates heterotypic transcription factor distribution and coordinates cardiogenesis. Cell164, 999–1014 (2016). ArticleCASPubMedPubMed CentralGoogle Scholar
- Zhang, J. et al. Harnessing of the nucleosome-remodeling-deacetylase complex controls lymphocyte development and prevents leukemogenesis. Nat. Immunol.13, 86–94 (2011). ArticlePubMedPubMed CentralCASGoogle Scholar
- Kanhere, A. et al. T-bet and GATA3 orchestrate Th1 and Th2 differentiation through lineage-specific targeting of distal regulatory elements. Nat. Commun.3, 1268 (2012). ArticlePubMedCASGoogle Scholar
- Garcia-Perez, L. et al. Functional definition of a transcription factor hierarchy regulating T cell lineage commitment. Sci. Adv.6, eaaw7313 (2020). ArticleCASPubMedPubMed CentralGoogle Scholar
Acknowledgements
The authors thank M. Romero-Wolf for helpful discussions and suggestions, and J. Ungerbäck, X. Wang, M. A. Yui and present and former members of the Rothenberg group, whose helpful discussion and published and unpublished data were important for the ideas in this Review. The authors apologize to colleagues whose relevant work could not be cited owing to space constraints. The authors gratefully acknowledge support from the Japan Society for the Promotion of Science KAKENHI (grant number JP19H03692), the Mochida Memorial Foundation for Medical and Pharmaceutical Research, the Naito Foundation and the Takeda Science Foundation (to H.H.), and from the US Public Health Service (R01AI135200, R01HL119102, R01HD100039 and R01HD076915) and the Albert Billings Ruddock Professorship (to E.V.R).
Author information
Authors and Affiliations
- Department of Immunology, Tokai University School of Medicine, Isehara, Kanagawa, Japan Hiroyuki Hosokawa
- Division of Biology and Biological Engineering, California Institute of Technology, Pasadena, CA, USA Hiroyuki Hosokawa & Ellen V. Rothenberg








